 
Therefore, there are various non-equivalent definitions of atomic radius. As you move down a group in the periodic table, electronegativity decreases due to the increase in the number of subshells and inner shells. 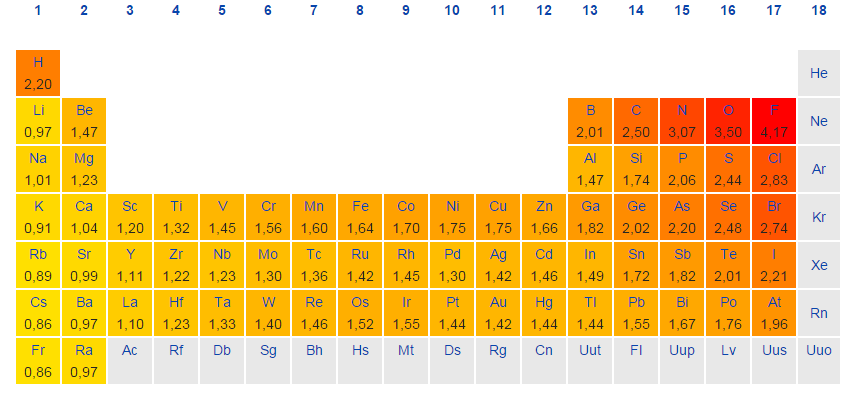
Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Palladium (Pd), Group 10, Atomic Number 46, d-block, Mass 106.42. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. The Naked Scientists Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. A modern version is shown in Figure 2.7.1 2.7. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. Current uses are as electrodes, heating elements and field emitters, and as filaments in light bulbs and cathode ray tubes. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. In contrast to its semi-mythical role in early metallurgy, these days the applications of tungsten are highly technological, making use of its hardness, stability and high melting point. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Hassium atom is 134pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The Royal Society of Chemistry's interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Scientists use the periodic table to quickly refer to information about an element, like atomic mass and chemical symbol. Left to right: Atomic number, Symbol, Name, Relative atomic mass, Electronegativity. Element Lead (Pb), Group 14, Atomic Number 82, p-block, Mass 207.2. Mass numbers of typical isotopes of Hassium are 269-271. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. Element Antimony (Sb), Group 15, Atomic Number 51, p-block, Mass 121.760. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Podcasts Produced by The Naked Scientists. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Hassium is a chemical element with atomic number 108 which means there are 108 protons in its nucleus. \).Atomic Number – Protons, Electrons and Neutrons in Hassium 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
